Vaccination via drinking water is one of the cornerstones of modern poultry production and accounts for the majority of immunization procedures carried out during the rearing and production cycles of commercial flocks. Although this method may appear straightforward, it actually involves a complex set of variables that can become potential causes of failure within a vaccination program. Water quality, which is often underestimated, and its comprehensive, end-to-end management are decisive factors in determining the effectiveness of live vaccines, directly influencing their viability and their uniform distribution across the farm.
Physicochemical parameters
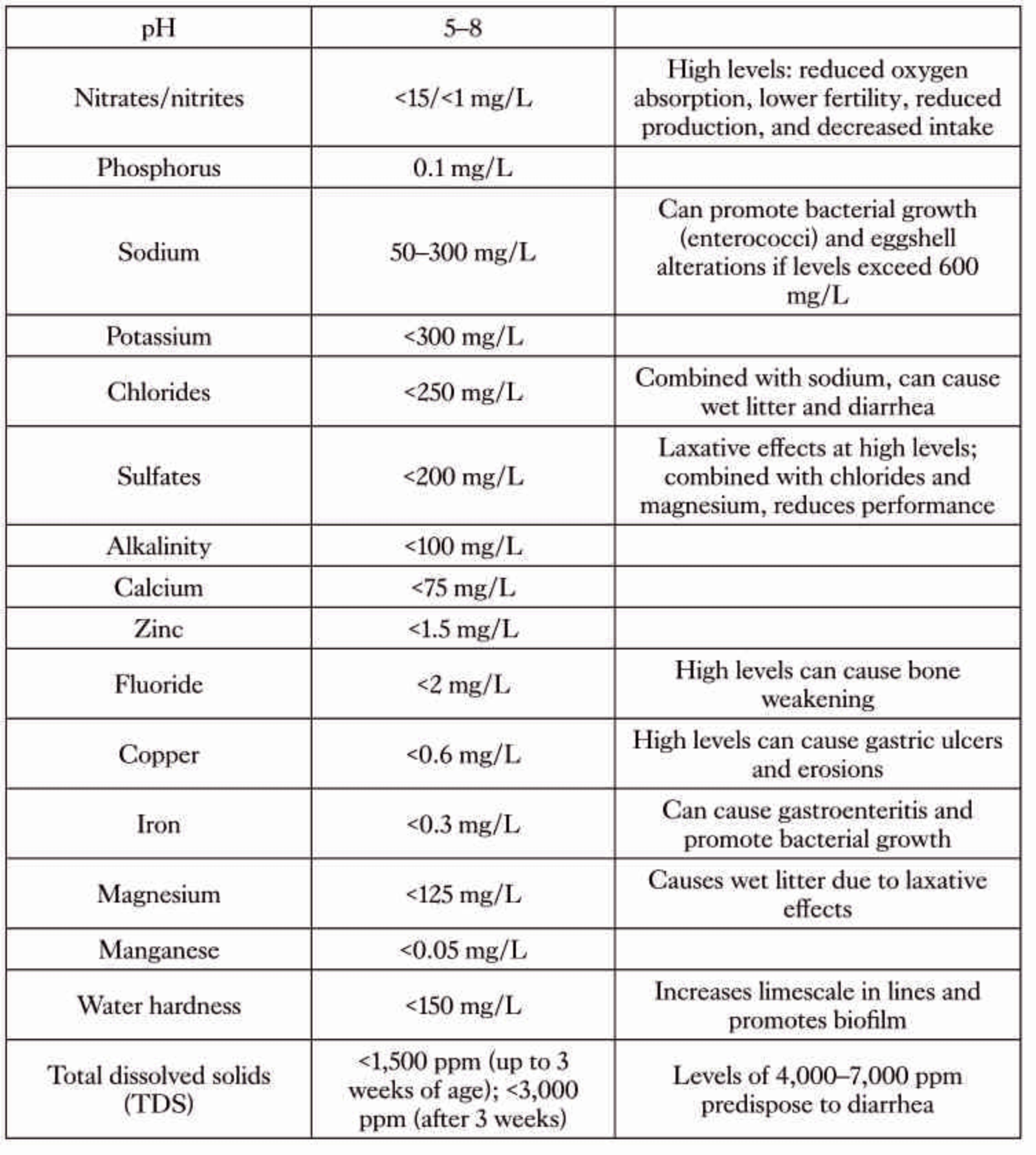
The water used for vaccination must meet specific parameters that often differ from those required for daily drinking water. Among all parameters, pH is the most critical factor: it must be maintained within the range of 6.5–7.8. Values outside this range can compromise the viability of live vaccines. Acidic pH values (<5) may, in some cases, make the administered water less palatable, discouraging intake, while alkaline pH values (>8.0) lead to the inactivation of these immunizing agents. Chlorine is one of the primary antagonists of live vaccines. Even minimal concentrations (0.1–0.2 ppm) of free chlorine exert bactericidal activity, while virucidal activity becomes evident at higher levels (0.3–0.5 ppm). Moreover, the organoleptic perception of chlorine (taste and odor) appears at levels above 0.5 ppm, serving as a reliable empirical indicator of lethality for most live vaccines. Heavy metals such as copper, iron, and manganese can form complexes with vaccine components, resulting in their inactivation. Water hardness, defined by the concentration of calcium and magnesium salts, must likewise be monitored to avoid interference with vaccine stability: it may contribute to scale formation within the lines, creating favorable conditions for microorganisms.
Among qualitative characteristics, turbidity is one of the most important parameters. When a sample is collected from the bottom of the drinking lines, it is visually assessed. Clear/transparent is the preferred condition, whereas flocculent material indicates poor quality. High degree of turbidity in drinking water negatively affects the animals’ immune response through inflammatory reactions and cell-mediated processes (Mohammed, 2008; Chen et al., 2018). Water temperature is also a relevant factor because bacterial replication increases above 25 °C (optimum 18–20 °C), negatively affecting the efficacy of applied treatments, including vaccinations.
Microbiological parameters and biofilm
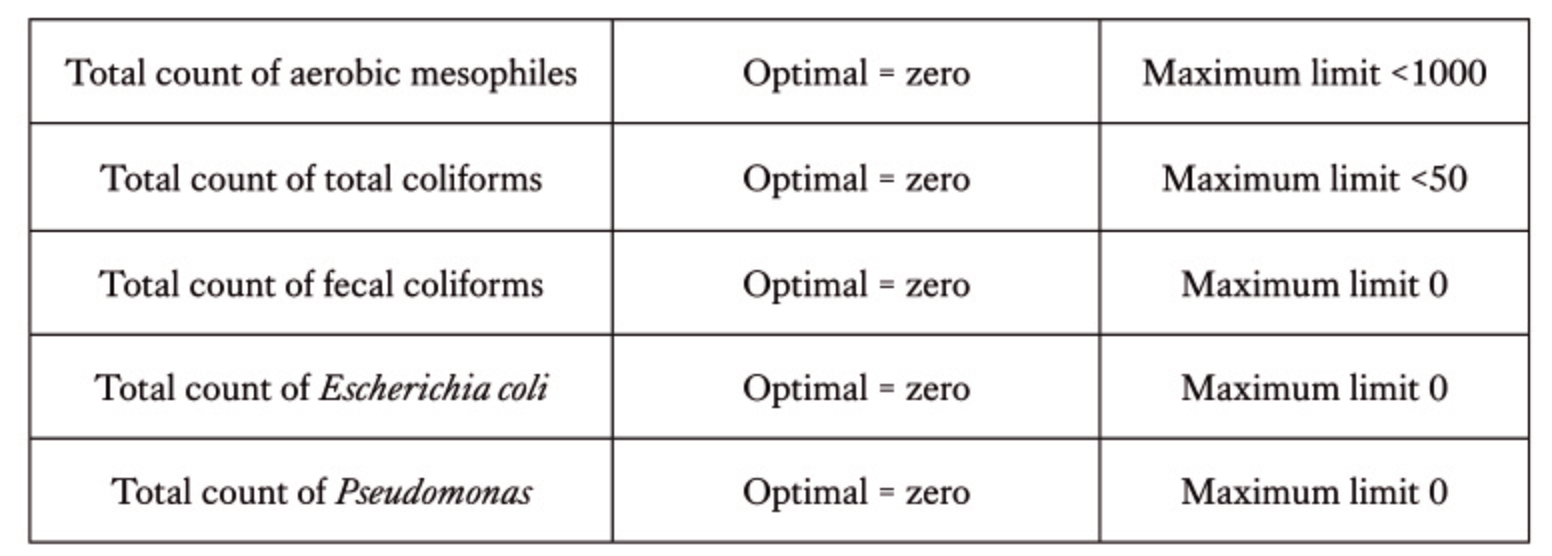
Biofilm in drinking water lines represents a major obstacle, frequently underestimated and undervalued to vaccine efficacy and effective immunization. These heterogeneous bacterial aggregates, composed of different microbial species, usually opportunistic like E. coli, Pseudomonas spp., Staphylococcus spp., Campylobacter spp., together with other organic contaminants (fungi, algae), settle on the inner surfaces of the pipelines, protected by a matrix of extracellular polymeric substances (EPS) as well as inorganic components (calcareous sources), factors that promote their stabilization.
Biofilm has multiple negative effects: it acts as a sink for vaccines on its surface, altering local pH and creating microenvironments unsuitable for the survival of immunizing antigens; it also reduces flow within the water system, increasing internal water pressure. Recent studies have shown that sessile bacteria embedded in biofilms develop resistance mechanisms to protect them from disinfectants and antimicrobials, rendering traditional sanitation protocols ineffective (Hahne et al., 2022).
Drinking systems: pre-vaccination checks and procedures
The design of drinking water distribution systems should include, upstream of the dosing pump, filters of approximately 80 microns (which can also serve multiple purposes such as absorption, sequestration, or mechanical filtration). These filters are installed to remove any suspended particles that might interfere with the correct distribution of vaccines through the system. During vaccination, however, all filters downstream of the dosing pump must be bypassed to prevent the accumulation of disinfectants and minerals on their surfaces.
The presence of dead spaces in the piping represents a critical risk factor. These areas can retain previously used disinfectant solutions, which — when mixed with the vaccine solution — compromise efficacy. It is also essential to design systems, where possible, with drain or purge valves at the ends of the lines to ensure they can be completely emptied prior vaccination.
Furthermore, the layout of the drinking lines and the water inlets within the system should be carefully assessed. Significant differences exist that may complicate uniform intake of the vaccine solution by the entire flock (for example, systems with central drops in the house versus those with only one inlet at the head, or multi-tier cage/aviary systems with specific animal arrangements). In such setups, depending on their design, the greatest risk is that animals closer to the water inlet may consume a larger volume of vaccine solution, while in some sections, particularly at the end of the line, the solution may not reach at all due to excessive water consumption in the initial stretch, possibly caused by over-settlement conditions.
Any biocides used (such as hydrogen peroxide, acidifiers, etc.) must be discontinued at least 24–48 hours before vaccination to allow complete removal of possible residues from the lines. High-pressure flushing of the lines can accelerate cleaning and/or emptying; performing this technique regularly (preferably once a week) also improves biofilm control by slowing its development. The effectiveness of these operations can be verified analytically using test strips to measure residual hydrogen peroxide and/or chlorine levels.
Finally, regular mechanical cleaning of nipples and cups (or bells, where used) with hot water and/or detergents (which must be thoroughly rinsed) helps remove organic residues from feces or litter, preventing local pH alterations and physical absorption of the vaccine used.
Quality control: systematic monitoring of drinking water
Analytical testing of drinking water should be performed at least once a year, with increased frequency during critical seasonal periods (summer and winter). Key parameters to assess include pH, chlorine, total hardness, heavy metals, total microbial load, and coliforms.
pH can be monitored using litmus paper and/or digital pH meters. Digital instruments are generally more sensitive and reliable if properly calibrated with the appropriate buffer solutions. In addition, commercially available digital probes allow continuous monitoring, providing real-time control of this parameter.
The uniform distribution of the vaccine solution throughout the drinking system lines can be verified using commercially available dyes or tracers (for example, methylene blue). Performing this test before vaccination helps identify areas where the solution might stagnate or fail to be evenly distributed, factors that could compromise the effectiveness of the vaccination procedure.
Moreover, drinking system pressure (approximately 1.5–2 bar) as well as flow rate (within the range of 50–80 ml/min) must ensure a constant supply throughout the entire system. Significant variations can result in over- or under-dosage, leading to uneven immunization within the flock and, in severe cases, possible reversion to virulence with adverse post-vaccination reactions (e.g., laryngotracheitis).
Optimization of vaccination procedures
Calculating the volume of water to be used requires specific knowledge of the farm’s drinking system as well as the flock’s water consumption. Based on these data, it is possible to determine water intake during the two hours following the morning feeding, which is the best time of day for vaccination (as a general empirical rule, this usually corresponds to 15–20% of the daily water intake). The volumes used must be adjusted according to several factors, primarily age, genetics, and ambient temperature.
The system’s dead space (any piping without usable bypasses/valves, recirculation tanks, length of pipeline from the dosing pump to the actual entry point into the drinking system, etc.) must be included in the total calculation to avoid unforeseen dilutions. Generally estimated at 10–15% (depending on the system), this volume can retain non-vaccine water and thus act as a dilution factor. Compensation for this residual volume can be achieved by proportionally increasing vaccine concentration or reducing the total dilution volume, ensuring a consistent dose-per-bird ratio.
The use of stabilizers is an essential component of vaccination via drinking water. These products contain active substances such as sodium thiosulfate, neutralize any residual chlorine, chelate heavy metals, and act as pH buffers, maintaining it within the optimal range. Skimmed milk powder (at a recommended rate of 2–3 grams per liter of water) is the traditional alternative to commercial stabilizers; milk proteins effectively bind chlorine and metal cations, protecting vaccines from inactivation. The stabilizing solution should be prepared at least 15–20 minutes before adding the vaccine to allow complete neutralization. Stabilizers may also be added during the pre-dilution step (demineralized water without stabilizer can be used as an alternative), in a smaller container together with the vaccine, as well as directly into the dosing pump tank.
 Vaccine reconstitution must take place in a controlled environment using disposable gloves and containers designated exclusively for this purpose (not previously used for disinfectant solutions or other products) in a suitable material (plastic). During preparation, exposure to UV light must be avoided, as UV radiation inactivates vaccines. Vials must be opened below the water level in the container used (containing at least 5–6 liters) to prevent airborne contamination and avoid potential loss of vaccine that could adhere to the container walls. Multiple rinsing of the vial (at least 2–3 times) with stabilized water ensures complete recovery of the vaccine content, which is particularly important for high-viscosity or adjuvanted vaccines.
Vaccine reconstitution must take place in a controlled environment using disposable gloves and containers designated exclusively for this purpose (not previously used for disinfectant solutions or other products) in a suitable material (plastic). During preparation, exposure to UV light must be avoided, as UV radiation inactivates vaccines. Vials must be opened below the water level in the container used (containing at least 5–6 liters) to prevent airborne contamination and avoid potential loss of vaccine that could adhere to the container walls. Multiple rinsing of the vial (at least 2–3 times) with stabilized water ensures complete recovery of the vaccine content, which is particularly important for high-viscosity or adjuvanted vaccines.
The optimal time of administration is early morning, starting at lights-on. In poultry, this corresponds to a peak in feeding activity and water consumption and takes advantage of natural behaviour to ensure rapid and uniform vaccine intake. Pre-vaccination water restriction of one to two hours stimulates thirst and concentrates intake of the vaccine solution into a short time frame. This restriction may be unnecessary if administration begins at lights-on, as the flock will already have undergone a minimum of eight hours of feed and water restriction. This restriction must be carefully evaluated in summer, under heat-stress conditions, to prevent potential adverse effects, particularly in laying birds (e.g. hyperthermia).
The recommended administration time window is generally an hour and a half to two hours, especially for more sensitive live viral vaccines. Shorter durations may result in incomplete vaccine coverage within the flock, whereas longer periods expose the vaccine to progressive inactivation. From a practical standpoint, it is advisable to divide the total vaccine dose into two equal phases of administration, each lasting an hour and a half to two hours; in the first phase, approximately 60% of the total dose is used, followed by a second phase delivering the remaining 40%. This helps less competitive birds also receive an adequate dose for immunization, a situation commonly observed in very long and/or multi-tier systems (e.g. aviary systems for laying hens).
Regular physical stimulation of the flock by the operator (at least every 30 minutes) plays an important role, as it encourages birds to move towards the drinking lines and supports uniform intake. Furthermore, if a dye is used, examining the oral cavity of birds sampled from different areas of the house becomes extremely useful. If at least 90% of birds show visible coloration of the tongue, the flock can be considered uniformly vaccinated.
Conclusion
Vaccination via drinking water in the poultry sector is a complex process that requires a multidisciplinary approach to ensure the effectiveness of immunizing agents. It is not merely a technical procedure, but the result of well-managed procedures in which every detail matters, from the chemical-physical and microbiological quality of the water to line cleaning and the proper preparation and administration of vaccines.
Only careful management based on rigorous protocols allows full exploitation of the advantages of drinking water prophylaxis. Systematic control of the parameters and procedures described not only guarantees vaccination effectiveness but also contributes to the farm’s economic sustainability.
Modern poultry farming therefore demands a rigorous scientific approach that integrates veterinary, engineering, and technical-management expertise to optimize this essential tool of preventive medicine.