
Mycoplasma gallisepticum (MG) and Mycoplasma synoviae (MS) remain persistent pathogens in poultry, causing respiratory disorders, synovitis, uneven growth, and reduced egg production. Determining the true source and timing of infection is critical for targeted interventions. Continuous monitoring using serology (ELISA) and molecular diagnostics (PCR and sequencing) enables differentiation between vertical transmission, hatchery contamination, and farm-level infection. Combining antibody kinetics and PCR results allows estimation of infection timing, improving decision-making for control measures. This article presents practical surveillance strategies, source analysis, and integrated control measures to sustain poultry production.
➤ Nader Rangsaz
Poultry Veterinarian (DVM), Faculty of Veterinary Medicine, Islamic Azad University, Shahrekord, Iran, Member of the Young Researchers Club, n.rangsaz@gmail.com
Introduction
MG and MS infections remain major challenges in commercial poultry. The production pyramid from GGP to broilers creates multiple points of potential contamination. A positive test alone does not indicate the source or timing of infection. Understanding whether infection originates from:
- upstream flocks (GGP/GP) > vertical transmission
- hatchery contamination > during incubation or handling
- farm-level infection > within the parent stock farm is essential for effective interventions, reducing unnecessary culling, and preserving production efficiency.
Economic impact in both breeders and broilers
MG and MS reduce growth rate, feed efficiency, egg production, and hatchability, leading to significant performance losses across the poultry industry. In broiler breeders, infection compromises reproductive efficiency, reduces egg quality, and lowers hatchability, while also producing weaker day-old chicks with reduced viability. These consequences not only decrease productivity but also may lead to trade restrictions, since certification programs often require Mycoplasma-free status. In broilers, infection is associated with airsacculitis, uneven growth, poor feed conversion, and higher carcass condemnations at processing plants. Although mortality may remain relatively low, the cumulative impact on flock uniformity and market weight is considerable. Importantly, co-infections with pathogens such as E. coli, Newcastle disease virus, or infectious bronchitis virus often exacerbate the clinical and economic effects of mycoplasmosis. Vertical transmission perpetuates infection down the production pyramid, while hatchery or farm-level contamination can trigger sporadic outbreaks. Therefore, accurate source tracking and infection timing estimation are crucial to minimize economic losses and to implement effective corrective measures.
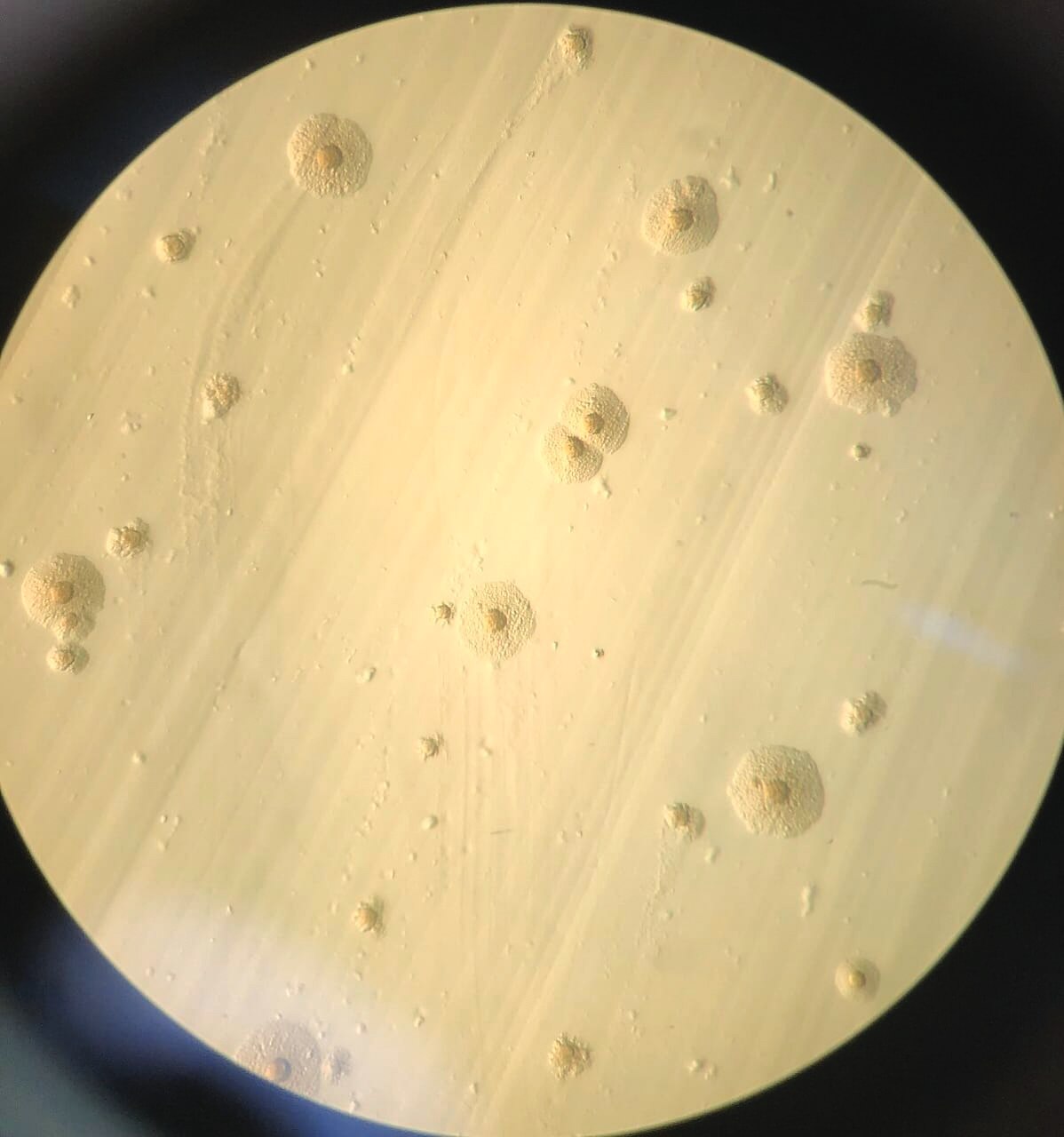
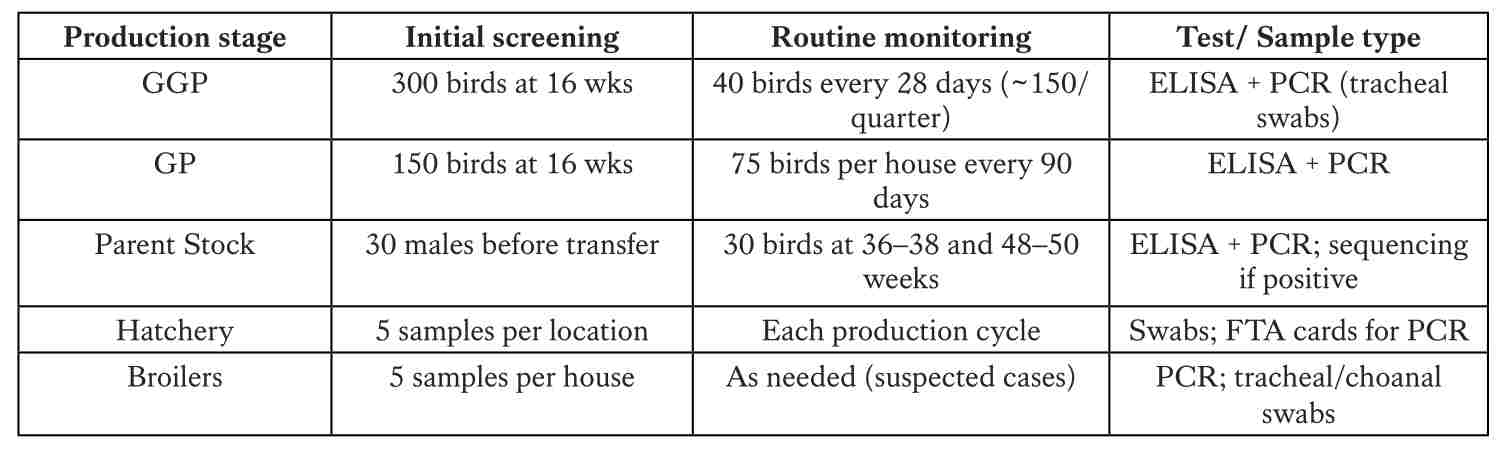
Monitoring plan and source analysis across the production pyramid
Monitoring should cover all pyramid levels, with key periods at pre-vaccination, pre-transfer, and peak production. Serology (ELISA) identifies immune response, PCR detects the pathogen, and sequencing confirms strain identity to determine the true source.
A structured monitoring program must align with the expected antibody response after vaccination. In practice, blood samples collected at different ages help confirm maternal antibody transfer. Sampling at around three weeks of age provides baseline data to document the natural decline in maternal antibodies. Baseline testing at 10 weeks ensures that the flock remains negative, while an additional test at 15 weeks, before transfer to the production site, confirms the negative status prior to movement.
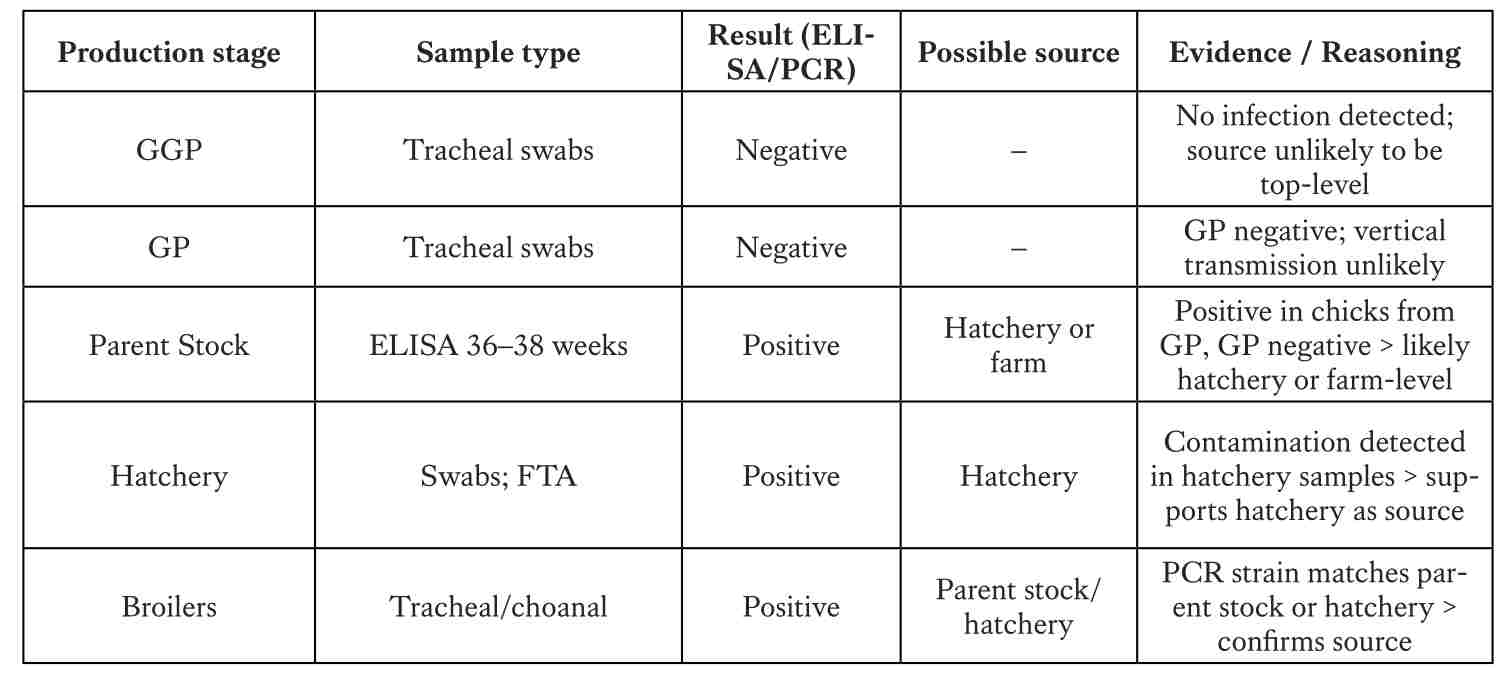
With vaccination commonly performed at six weeks, marking the start of active immunity, post-vaccination monitoring at 21 weeks, prior to peak lay, is critical. Using ELISA, typical titers following MG F-strain vaccination range between 2,000–8,000, while ts-11 usually produces 1,000–3,000 (30–70% positives). For MS-H vaccine, mean titers are expected in the range of 500–3,000 without wild challenge. Any values significantly above these levels (e.g. >5,000–23,000 with 90–100% seropositive samples) strongly suggest field infection rather than vaccine response. Additional monitoring at 32 weeks (to detect breakthrough infections), 44 weeks (to evaluate vaccine duration), and 55 weeks (for end-of-lay cycle status) provides mid- and late-production surveillance, ensuring that no breakthrough infections occur. This approach allows managers to clearly separate normal vaccine serology from true Mycoplasma challenge and to implement corrective actions in time.
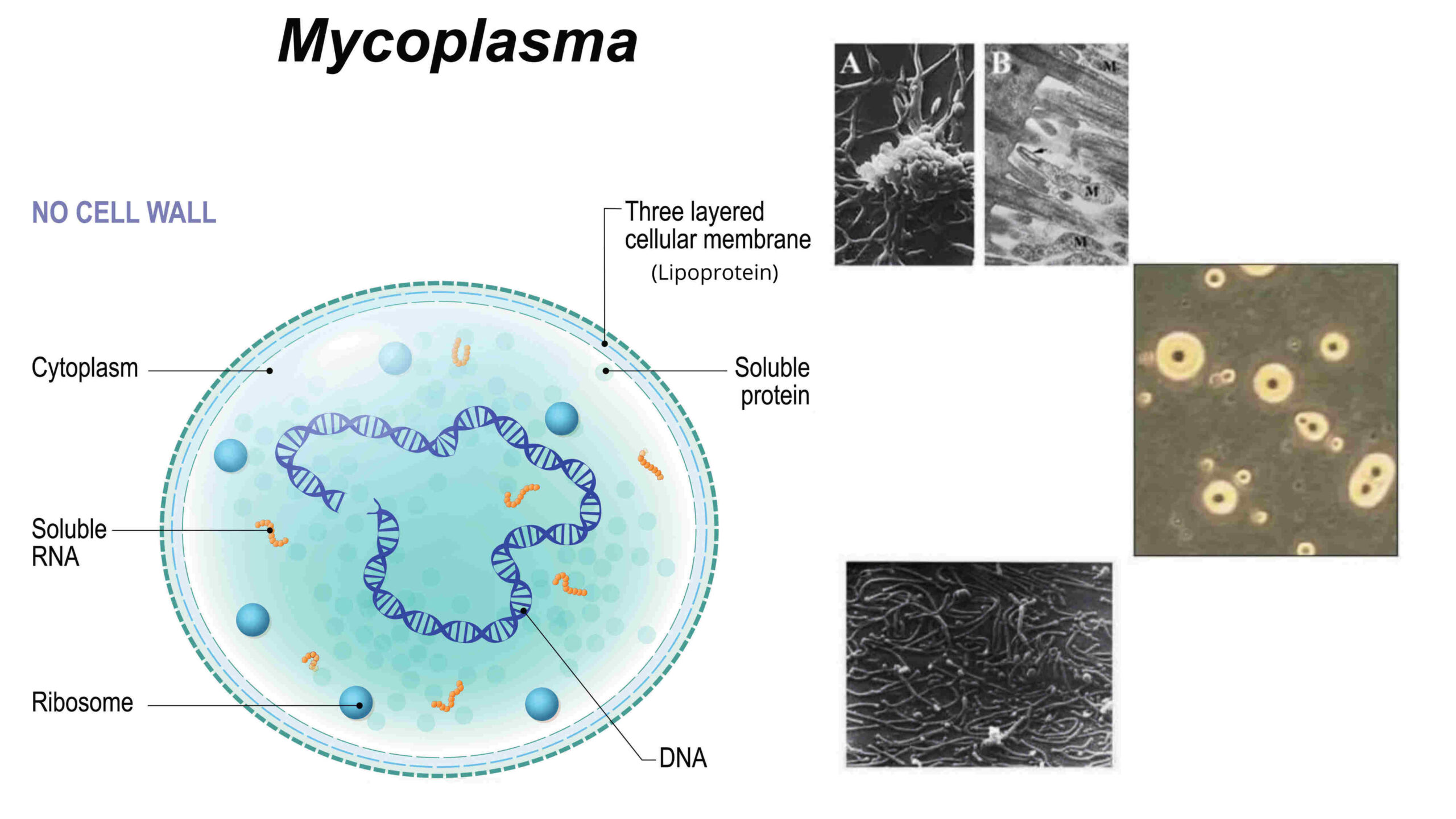
Monitoring for antibodies must be carried out prior to vaccination with live vaccines.
Enzyme-linked immunosorbent assays can be used to differentiate vaccinated flocks from those undergoing a challenge.
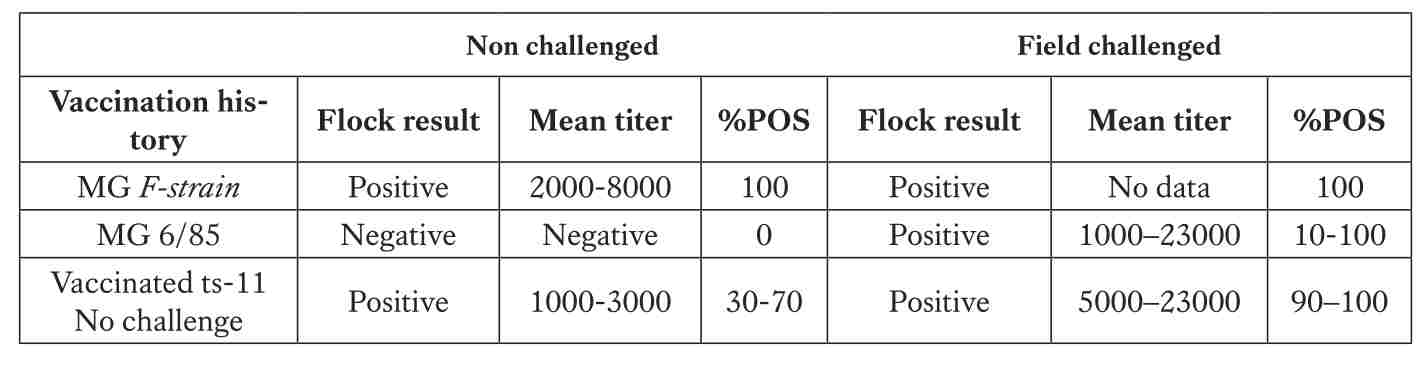
*A titer of ≥ 668 is positive.
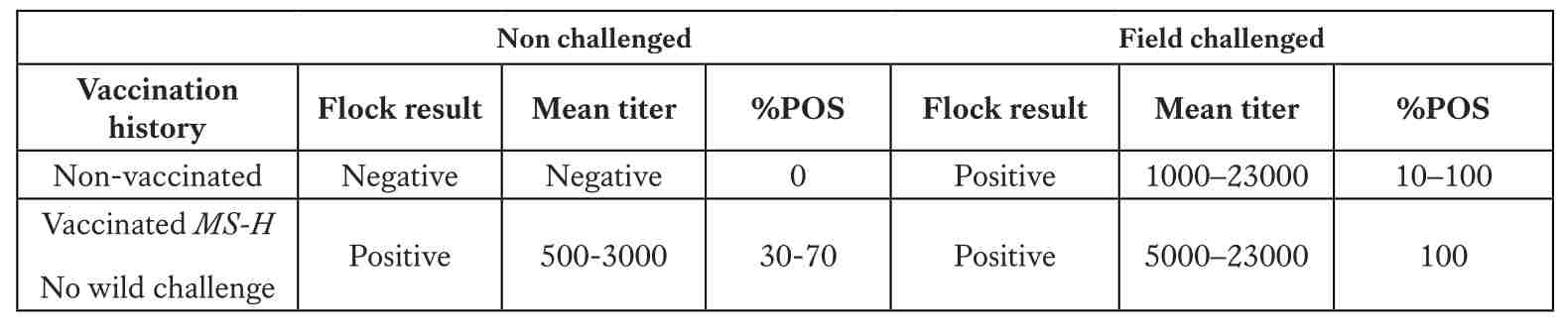
*A titer of ≥ 668 is positive.
Timing of infection using antibody kinetics
Key points
- Maternal antibodies decline over time; a positive titer at day 1 reflects maternal transfer;
- a rising titer after the decline indicates natural infection; the slope helps estimate infection timing;
- comparison with vaccination differentiates vaccine response from natural infection;
- serial sampling is required for precise estimation;
- PCR confirmation supports timing estimate and identifies the strain.
Diagnostic approaches
- ELISA: large-scale flock screening;
- PCR: rapid detection and strain differentiation;
- culture and immunofluorescence: gold standard, but it is costly and slow (up to 4 weeks), typically reserved for certification and research.
At least two independent positive results are recommended for confirmation; titers > 1:80 (or kit cut-off) indicate infection.
Control and prevention
- Elimination: cull positive breeders to prevent vertical spread;
- vaccination: live (F, 6/85, TS-11, and MS-H) or inactivated vaccines; PCR differentiates vaccine from field strains;
- antibiotics: mycoplasmas are generally susceptible to macrolides, tetracyclines, fluoroquinolones and the combination of lincomycin and spectinomycin to reduce clinical signs;
- biosecurity and sourcing: hygiene, audits, certified Mycoplasma-free source;
- continuous monitoring: ensures early detection and source identification.

Conclusion
Identifying the true source and approximate timing of infection is critical. Combining ELISA kinetics, PCR, sequencing, clinical observation, and necropsy allows differentiation between vertical, hatchery, and farm-level infection. Accurate source and timing identification prevent unnecessary culling, focuses interventions, and improves flock sustainability.
MG and MS threaten flock health and productivity. Continuous monitoring, molecular and serological diagnostics, biosecurity, and vaccination are essential. Source identification and timing estimation enable targeted control, reducing economic losses and sustaining poultry production.
References
Achari, R., & Morrow, C. (2018). Diminishing Control of Avian Mycoplasmas. Association of Avian Health Professionals, India.
BioChek. (s.d.). Live Mycoplasma Vaccines and the Use of Monitoring: Interpretation of BioChek MG ELISA titers 6–12 weeks post vaccination with Live MG vaccines. BioChek Application PDF.
Ferguson-Noel, N. (2014). Control of Avian Mycoplasmosis. The Poultry Informed Professional, University of Georgia.
Kleven, S.H. (2000). Mycoplasma Update. The Poultry Informed Professional, University of Georgia.
Morrow, C.J. (2017). Practical Mycoplasma Control for Poultry Production in Asia. International Production Poultry, 25(1), 35–37.
Rosales, A.G. (2019). Mycoplasmosis Prevention and Control in Broiler Breeders and Broilers. Aviagen.








