
Immune activation is the first stage of the immune response necessary to protect the body. It is accompanied by significant metabolic costs and inflammation. When immune cells are activated, their metabolism is reprogrammed — a large-scale shift in energy and nutrient use to meet increased demands for protein, lipid, and nucleic acid synthesis. This metabolic shift reduces growth performance and feed conversion efficiency.
Field observations
Comparison of production data shows major differences in broiler performance even under the same genetics, feed, and housing conditions. These differences are directly related to the level of immune load.
When vaccination programs are intensified or infection pressure is high, growth rate and feed conversion ratio (FCR) decline. In contrast, in New Zealand, where broilers are raised under minimal infectious pressure, results are outstanding: at 34 days of age, body weight reaches 2,600 g, FCR is 1.29, and livability is 98%.
The difference is not due to genetics or feed formulation, but to the level of immune load. When the immune system is at rest, all nutrients can be directed toward growth rather than defense.
The dual nature of immunity
The immune response consists of two interconnected arms: innate and adaptive immunity.
Innate immunity is the first line of defense, based on phagocytosis, cytokine release, complement activation, and inflammation. It develops within hours but is very energy-demanding: energy consumption rises by 5–10%, protein catabolism increases, and body temperature rises.
Adaptive immunity develops more slowly: a full T- and B-cell response may take up to two weeks. It is more specific and less energy-intensive. Once immune memory is established, secondary responses require minimal energy.
Live vaccines trigger the same immune-metabolic cascades as field viruses, but the response is milder and causes less loss of productivity. This allows adaptive immunity to form with minimal reduction in growth and energy efficiency.
Phases of the immune response in broilers
Recognition and innate response (0–24 h)
Macrophages and heterophils recognize pathogens through TLR receptors and release pro-inflammatory cytokines (IL-1β, IL-6, TNF-α). Feed intake decreases, body temperature rises, and the liver increases synthesis of acute-phase proteins. NF-κB and JAK-STAT signaling pathways are activated, increasing energy use for inflammation.
Adaptive response (2–7 days)
Lymphocyte proliferation, antibody production, and memory cell formation begin. The demand for arginine, glutamine, threonine, and nucleotides rises — they serve both as building blocks and energy sources for immune cells. The liver remains active in producing acute-phase proteins, reducing the nutrients available for growth.
Resolution and recovery (7–14 days)
Anti-inflammatory cytokines IL-10 and TGF-β are activated, reactive oxygen species decrease, antioxidant balance is restored, and anabolic pathways (mTOR, IGF-1) are reactivated.
Under repeated vaccinations or concurrent field infections, this phase may be prolonged, leading to chronic catabolic states and oxidative stress.
Each phase has specific metabolic priorities:
- Innate: glucose and antioxidants
- Adaptive: amino acids and nucleotides
- Recovery: lipids and sulfur-containing amino acids
Managing chronic inflammation and supporting the immune system
The avian immune system consists of physical barriers and cellular mechanisms that protect against pathogens.
Inflammation is a vital part of innate immunity, but chronic activation is costly and reduces productivity. Effective immune regulation helps limit inflammation and preserve nutrients for growth.
Role of epithelial health
The health of epithelial tissues is key to balanced immune function. The gastrointestinal and respiratory epithelia act as the first barrier against infections. Stress factors such as heat, mycotoxins, or electrolyte imbalance can disrupt tight junctions, causing chronic inflammation and increased intestinal permeability.
The cost of immune activation
Activation of innate immunity requires large amounts of amino acids, energy, and trace minerals. During chronic inflammation, nutrients are diverted from growth toward immune processes, worsening FCR and body weight gain.
Cytokines IL-1β, IL-6, and TNF-α activate NF-κB and STAT3 pathways, shifting metabolism from growth to defense.
Experimental immune stimulation (LPS challenge or vaccination) increases maintenance energy needs by 5–10% and reduces protein synthesis. Liver metabolism shifts toward catabolism of branched-chain amino acids, mTOR activity decreases, corticosterone levels rise, and tissue insulin sensitivity declines.
Body weight can drop by 10–30%, and FCR worsens as nutrients are redirected to cytokine, antibody, and acute-phase protein synthesis. Even after inflammation resolves, the effects can persist for several days, explaining temporary “growth dips” after vaccinations.
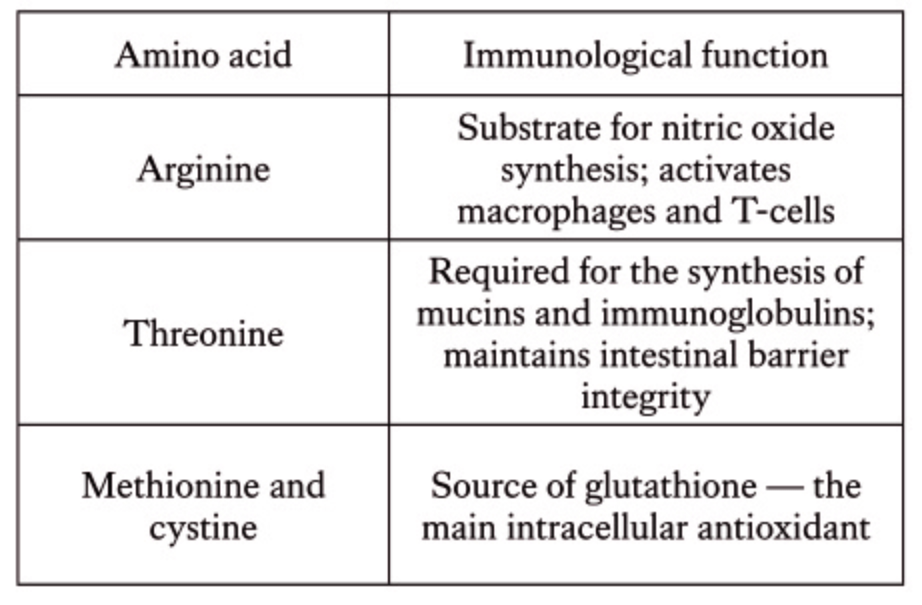
The demand for arginine and threonine increases by 10–15%, and for methionine and cystine by about 5%. Maintaining optimal ratios of these amino acids to lysine is critical for sustaining performance under immune load.
Antioxidants and immune homeostasis
Immune activation increases oxidative stress, especially in the intestinal mucosa. Adequate antioxidant supply shortens the inflammatory phase and speeds up recovery.
- Vitamin E and selenium increase glutathione peroxidase activity and antibody levels.
- Vitamin C lowers corticosterone concentration and supports phagocytosis.
- Postbiotics and paraprobiotics reduce pro-inflammatory cytokines and raise IL-10, improving nutrient absorption.
- Early microbiota modulation enhances intestinal immune development and NK-cell activity.
Nutritional strategies duringimmune activation
When the immune system is activated, requirements for nutrients, energy, and antioxidants increase, requiring diet adjustments:
- Add antioxidants to neutralize free radicals and reduce oxidative stress.
- Increase levels of key amino acids (arginine, threonine, methionine, and cystine) to support immune protein synthesis and tissue repair.
- Raise metabolizable energy (ME) levels.
Field observations conducted by the author in commercial broiler operations demonstrated the effectiveness of compensating immune costs through nutrition.
In one experiment, chicks from the same breeder flock were placed in identical houses. The site was known to have circulating IBD, IBV, NDV, Reovirus, and low-pathogenic avian influenza.
The control group received a standard diet according to breed recommendations. The test group received higher ME levels and increased threonine and methionine.
At the end of the trial, the experimental group showed:
- +4.3 g/day higher average daily gain;
- 6.2% higher livability;
- 0.08 kg lower FCR per kg of body weight compared to the control.
Conclusion
Immune activation is an unavoidable response of the immune system to pathogens or vaccines. However, its intensity determines how deeply it affects energy and amino acid metabolism.
Under mild or moderate immune activation, productivity losses can be compensated through proper feeding strategies, by increasing dietary energy, enhancing antioxidant protection, and optimizing amino acid profiles (arginine, threonine, methionine, cystine).
However, under heavy immune load (frequent vaccinations, field virus exposure, or poor biosecurity) the effectiveness of nutritional compensation drops sharply. Even with higher dietary energy and amino acid levels, growth rate and FCR cannot return to normal, as much of the nutrients are diverted to chronic inflammation and immune protein synthesis suppression.
Therefore, maintaining strict biosecurity is essential for economic efficiency. Key measures include:
- controlling farm access and maintaining sanitary barrier
- thorough cleaning and disinfection
- optimizing vaccination programs based on maternal immunity, local disease pressure, and vaccine compatibility
In the future, accounting for the nutritional requirements of the immune system should become a standard component of precision poultry nutrition.
Bibliography
- Aguzey, H. A., Gao, Z., Haohao, W., Guilan, C., Zhengmin, W., Junhong, C., & Zhi Li, N. (2020). The role of arginine in disease prevention, gut microbiota modulation, growth performance and the immune system of broiler chicken – a review. Annals of Animal Science, 20(2), 325–341.
- Ahiwe, E. U., Omede, A. A., Abdallh, M. B., & Iji, P. A. (2016). Managing dietary energy intake by broiler chickens to reduce production costs and improve product quality. In InTechOpen Book Chapter.
- Dadfar, M.-J., Vaez Torshizi, R., Maghsoudi, A., Ehsani, A., & Masoudi, A. A. (2023). Trade-off between feed efficiency and immunity in specialized high-performing chickens. Poultry Science.
- Hollemans, M. S., de Vries Reilingh, G., de Vries, S., Parmentier, H. K., & Lammers, A. (2020). Effects of early nutrition and sanitary conditions on oral tolerance and antibody responses in broiler chickens. Veterinary Sciences, 7(4), 1–12.
- Hu, W., Du, L., Shao, J., Qu, Y., Zhang, L., Zhang, D., Cao, L., Chen, H., & Bi, S. (2024). Molecular and metabolic responses to immune stress in the jejunum of broiler chickens: transcriptomic and metabolomic analysis. Poultry Science.
- Li, R. F., Liu, S. P., Yuan, Z. H., Yi, J. E., Tian, Y. N., Wu, J., & Wen, L. X. (2023). Effects of induced stress from the live LaSota Newcastle disease vaccination on the growth performance and immune function in broiler chickens. Poultry Science.
- Liu, K., Zhen, W., Bai, D., Tan, H., He, X., Li, Y., Liu, Y., Zhang, Y., Ito, K., Zhang, B., & Ma, Y. (2023). Lipopolysaccharide-induced immune stress negatively regulates broiler chicken growth via the COX-2–PGE2–EP4 signaling pathway. Frontiers in Immunology.
- Liu, L., Qin, D., Wang, X., Feng, Y., Yang, X., & Yao, J. (2015). Effect of immune stress on growth performance and energy metabolism in broiler chickens. Food and Agricultural Immunology, 26(2), 194–203.
- Maroufyan, E., Kasim, A., Hashemi, S. R., Loh, T. C., Bejo, M. H., & Davoodi, H. (2010). The effect of methionine and threonine supplementations on immune responses of broiler chickens challenged with infectious bursal disease. American Journal of Applied Sciences, 7(1), 44–50.
- Sheikh, I. S., Bajwa, M. A., Rashid, N., Mustafa, M. Z., Tariq, M. M., Rafeeq, M., Samad, A., Asmat, T. M., & Ullah, A. (2020). Effects of immune modulators on the immune status of broiler chickens. Pakistan Journal of Zoology, 52(3), 1095–1100.
- Yang, J., Liu, L., Sheikhahmadi, A., Wang, Y., Li, C., Jiao, H., Lin, H., & Song, Z. (2015). Effects of corticosterone and dietary energy on immune function of broiler chickens. PLOS ONE, 10(3), e0122004.
- Ye, J., Yang, H., Hu, W., Tang, K., Liu, A., & Bi, S. (2023). Changed cecal microbiota involved in growth depression of broiler chickens induced by immune stress. Poultry Science.
- Zheng, A., Zhang, A., Zheng, Z., et al. (2021). Molecular mechanisms of growth depression in broiler chickens (Gallus gallus domesticus) mediated by immune stress: A hepatic proteome study. Journal of Animal Science and Biotechnology, 12(90).








