
Avian reoviruses (ARVs). They are non-enveloped viruses of 70-80 nm size having an icosahedral symmetry. The viral genome has 10 segments of double stranded RNA contained inside a double-shelled arrangement of capsid proteins. The ARVs infect several different avian species and are named accordingly viz. chicken reovirus (CRV), duck reovirus (DRV), goose reovirus (GRV), and turkey reovirus (TRV).
These viruses are responsible for inapparent infections as well as frank disease in birds including enteritis, hepatitis, neurological disease, myocarditis, respiratory distress and viral arthritis/tenosynovitis. Severity and clinical manifestation of the disease often depend on age and immune status of the host, virus pathotype, and route of exposure (oral, intratracheal, footpad, or subcutaneous).
Turkey arthritis reovirus (TARV) was isolated in 1980s from joints and tendons of turkeys showing lesions of arthritis and tenosynovitis but the disease could not be reproduced experimentally. Since then, there were no reports of viral arthritis in turkeys until 2011, when the condition re-emerged in midwestern and eastern states of the US.
Diagnostic case definition of turkey reoviral arthritis
Porter (2017) defines turkey reoviral arthritis as a progressive condition that appears as early as 10-12 weeks of age in male, and sometimes female, commercial turkeys. Signs are most severe when the birds reach 15-16 weeks of age. Clinical signs include reluctance to move, recumbency, and limping of one or both legs. There is often uni- or bilateral swelling of the hock (intertarsal) joint. Lesions in acutely affected birds at necropsy are uni- or bilateral enlargement (subcutaneous edema) of the hock joints, which contain increased volume of clear yellow to serosanguinous synovial fluid. Such fluid can expand the sheath of the gastrocnemius and digital flexor tendons. In chronic cases, there is bruising of the skin of the hock with prominent periarticular fibrosis, edema and occasional large flecks of fibrin within the subcutis and tendon sheaths. In severe cases, rupture of gastrocnemius and/or digital flexor tendon is observed along with hemorrhage at the level of the rupture. The virus can affect 35-70% in a flock, leading to huge economic losses due to poor weight gain, uneven growth, poor feed conversion, increased mortality, and reduced market weight of commercial turkeys. The affected birds cannot walk to the waterers and feeders and hence the disease has animal welfare concerns. Histological sections of gastrocnemius tendon and sheath reveal lymphocytic infiltrates in the sub-synovium in acute cases, progressing to prominent sub-synovial and peritendon fibrosis in chronic cases. Secondary bacterial infections (e.g., Staphylococcus) occasionally occur and are accompanied by heterophilic inflammation.
Diagnosis
Arthritis/tenosynovitis caused by TARV is diagnosed on the basis of history, typical clinical signs, gross and microscopic lesions, virus isolation, and RT-PCR. Definitive diagnosis requires ruling out other causes of lameness in turkeys, e.g., osteomyelitis, primary bacterial arthritis, muscle rupture, footpad dermatitis, and synovitis due to Mycoplasma. Isolation of TARV from gastrocnemius and/or digital flexor tendon in embryonated eggs or cell culture confirms the etiology of the disease. Additionally, serology, electron microscopy and molecular assays are available for detection of TARV infection.
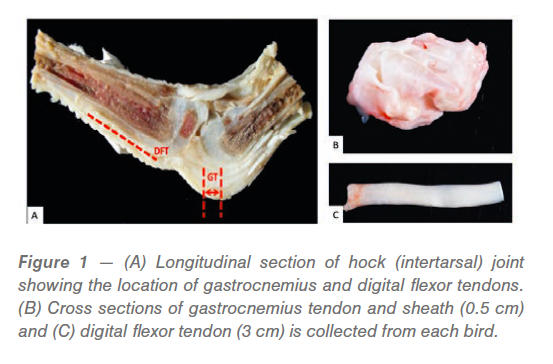
Sample collection and processing
The affected leg is sectioned longitudinally, and a 0.5 cm cross section of gastrocnemius tendon (GT) and 3 cm long piece of digital flexor tendon (DFT) is cut and sampled (Figure 1). Tendon samples from 3-6 affected legs are collected and pooled followed by homogenization in Genogrinder tubes containing phosphate buffered saline (PBS). The Genogrinder tubes are subjected to three cycles of vigorous shaking at 1200 strokes/min for 4 min each to homogenize the tendon samples (Figure 2). The homogenate is centrifuged at 6000 xg for 10 min followed by the collection of the supernatant, which is then used for virus isolation and/or rRT-PCR. Virus isolation can be done either in embryonated chicken eggs or cell cultures.

Virus isolation in embryonated eggs
Isolation of TARV is best achieved by inoculation of the supernatant from tendon homogenate in 6-day-old, Specific Pathogen Free (SPF) embryonated chicken eggs via the yolk sac route. Virulent virus generally causes embryo mortality within 5-6 days of inoculation. The eggs are incubated for 5-6 days and then chilled in the refrigerator for at least 4 hours. The yolk sac and embryos with lesions are harvested, subjected to 3 freeze thaw cycles, and then homogenized in a Stomacher. Following centrifugation at 6000 xg for 30 min, the supernatant is collected and inoculated in Japanese quail fibrosarcoma cell line (QT-35) cells.
Virus isolation in cell cultures
Supernatant collected from embryonated eggs or supernatant from homogenized tendons is inoculated in monolayers of QT-35 cells at 80-90% confluency followed by incubation in a 5% CO2 incubator at 37 ºC. The cells are observed daily for the appearance of virus-induced cytopathic effects (CPE), which include syncytium formation and detachment of cells within 48-72 hours of incubation. Once CPE is observed, the infected cells are subjected to three freeze thaw cycles followed by centrifugation at 6000 xg for 10 min. The supernatant is collected for further processing. The identity of the virus can be confirmed by immunofluorescence or rRT-PCR.
Fluorescent antibody test (FAT)
The cells showing CPE are scraped, placed on a glass slide, and fixed in acetone. A drop of antireoviral polyclonal antiserum conjugated with fluorescein isothiocyanate (FITC) is added to the fixed cells and incubated for 2 hours in a 5% CO2 incubator at 370C. The cells are counter stained in Evans Blue and then examined under a fluorescent microscope. Samples showing fluorescence are considered positive for reovirus.
Molecular assays
We have developed a one-step real time RT-PCR (qPCR) for the detection of TARV using TARV-specific S1 and/or S3 primers (manuscript in preparation). A universal avian reovirus qPCR for the detection of all avian reoviruses has also been developed. Viral harvest from infected cell cultures and embryonated chicken eggs can be confirmed by rRT-PCR and/or RT-PCR.
Whole genome sequencing (WGS) by Next generation sequencing (NGS)
Whole genome sequencing is a newer technique that can detect the whole genome of a given virus. Initially, this techniques was used as a research tool but is now being used relatively routinely in the diagnostic labs.
Serological assays
Serological methods such as enzyme linked immunosorbent assay (ELISA) and serum neutralisation (SN) are available to detect antibodies to avian reoviruses.
ELISA
Currently, commercial ELISAs are available for the detection of antibodies against chicken reoviruses but no turkey-specific ELISA exists at this time. Chicken-specific ELISA is sometimes used in the field to monitor antibody titers in breeder turkeys that are vaccinated with autogenous TARV vaccines and the offspring from those breeders. The presence of high antibody titers in birds is used as an indication of field challenge by turkey producers. This gives the turkey producers time to alter their processing schedule and salvage some birds. We have developed an ELISA for the detection of TARV antibodies. Preliminary results indicate that this ELISA is as sensitive as the commercially available chicken-specific ELISA.
Serum Neutralization test
In addition to ELISA, serum neutralization (SN) can also be used for detecting seroprevalence of TARVs. In addition, cross neutralization assays can be used to characterize newly isolated TARVs and determine their antigenic relationships.
Conclusions
Viral arthritis/tenosynovitis caused by TARV is diagnosed on the basis of history, typical clinical signs, gross and microscopic lesions supported by laboratory techniques like virus isolation, and RT-PCR where virus isolation (VI) is considered as a gold standard test for the diagnosis of avian reovirus. Additionally, serology, electron microscopy and molecular assays are available for detection of TARV infection. With the advent and advancement of sequencing technologies, rapid diagnosis and molecular characterization of TARVs is possible with more precision.
References are available on request
From the Proceedings of the 68th Western Poultry Disease Conference (2019)








